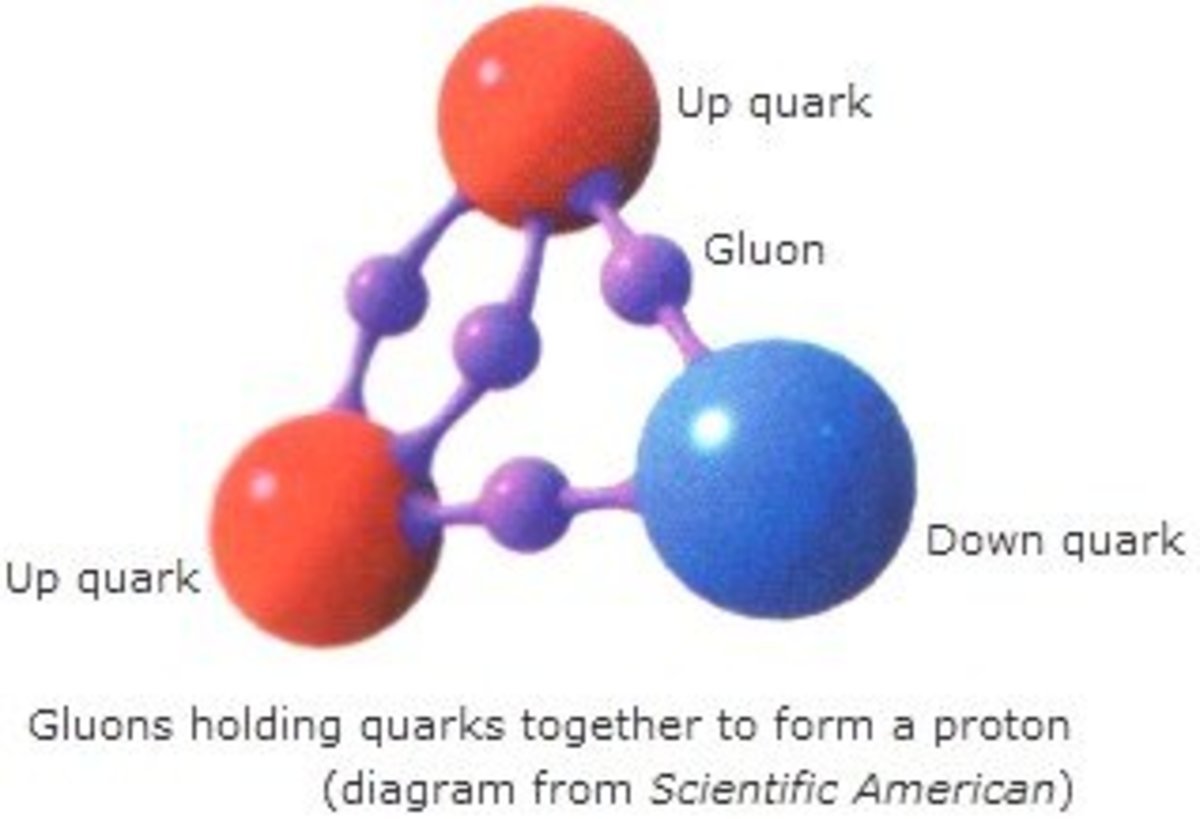
Among these subatomic particles, a proton has +1 charge, and an electron has -1 charge while neutron has no charge. Around 99% of the atom’s mass is centred in the nucleus because the mass of an electron is almost negligible. Typically, the number of neutrons, protons and electrons are equal to each other, but in the case of isotopes, the number of neutrons is different from that of protons. 
The atomic nucleus is made of protons and neutrons (and there are some other subatomic particles as well). When considering the structure of an atom, it contains a nucleus and electrons moving around the nucleus. These are very minute units typically the size is around 100 picometers. All gases, solid matter, liquids and plasma contain atoms. Therefore, it is the smallest unit of matter, and a certain atom represents the properties of the chemical element to which it belongs. Side by Side Comparison – Atoms vs Particles in Tabular FormĪtoms are the smallest particles of a chemical element that can exist. However, the term particle in chemistry refers to any small localized object which has physical properties such as volume, density and mass. But according to recent research studies, the atom is made of several small particles called subatomic particles. In the past, people thought that the atom was the smallest thing that exists and we cannot break it down further. The key difference between atoms and particles is that atoms are small units made of several particles, whereas particles are minute portions of matter.Īn atom is the smallest unit of all matter.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
